SNF472 selectively inhibits pathological calcification by binding to hydroxyapatite crystals.
Palma, Spain and San Diego, USA, June 25, 2020 – Sanifit, a clinical-stage biopharmaceutical company focused on developing treatments for calcification disorders, announced today that data examining the mechanism of action of SNF472, Sanifit’s lead compound in development. has been published in the scientific journal British Journal of Pharmacology (BJP).
SNF472 is a novel calcification inhibitor for the treatment of cardiovascular calcification (CVC) and the rare disease called calciphylaxis in patients undergoing hemodialysis. SNF472 is currently in clinical phase 3 for the treatment of calciphylaxis.
In dialysis patients, vascular calcification occurs when calcium and phosphate aggregate into hydroxyapatite (HAP) crystals and deposit on arterial walls. The PAH crystals block small arteries in the skin and soft tissues. SNF472 binds with high affinity to PAH crystals, the common final stage of CVC processes, and selectively inhibits their accumulation in arterial walls regardless of the etiology of PAH formation.
The studies examined the binding characteristics of SNF472 on PAH crystals in a variety of in vitro and in vivo models. Binding was found to be rapid (80% within 5 minutes) and unsurpassed. SNF472 inhibited PAH crystal formation with an EC50 of 3.8 μM, with complete inhibition at 30.4 μM. At the lowest concentration tested (1 μM) in vascular smooth muscle cells, SNF472 inhibited calcification by 67%, with complete inhibition at 30 μM. Interestingly, the mean plasma concentration in an open-label phase 2 study with SNF472 in patients with calciphylaxis was 28 μM after 12 weeks of treatment. In that study, improvements in wound healing (primary endpoint), pain and quality of life (secondary endpoints) were observed. Similar concentrations significantly attenuated the progression of coronary arterial calcification in a phase 2b, double-blind, placebo-controlled trial in hemodialysis patients.
SNF472 showed no deleterious effects on bone mineralization in dogs after 9 months at 25 mg/kg (maximum concentration 384 μM) and the EC50 for calcium chelation in vitro is 539 μM. Full results can be accessed online here.
These data support the continuation of clinical investigation of SNF472 for the treatment of vascular calcification and calciphylaxis in patients undergoing dialysis.
Joan Perelló, CEO of Sanifit, commented, “We are very pleased with the publication of these data by BJP, which provide valuable information on the mechanism of action of SNF472 and its significant potential as an effective inhibitor of PAH crystallization in the cardiovascular system. With no currently approved therapy for vascular calcification or calciphylaxis, which are associated with increased mortality and morbidity in patients undergoing dialysis, we look forward to advancing SNF472 in our clinical development for patients in need.”
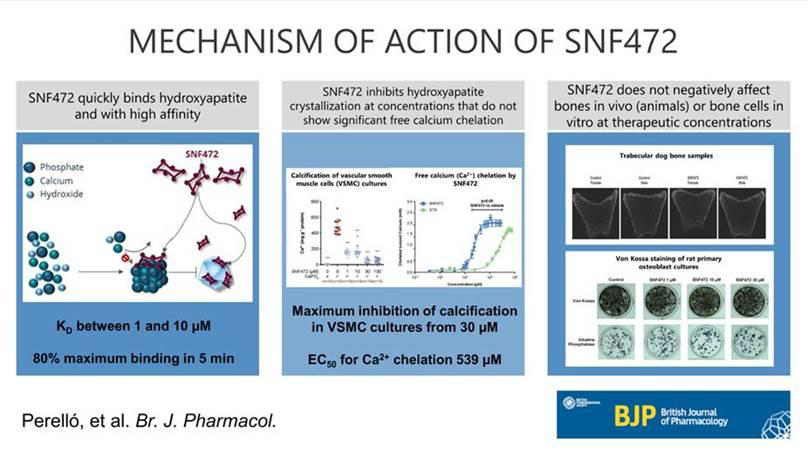
Figure 1: Graphical abstract of the article: Perelló, J, Ferrer, MD, del Mar Pérez, M, et al. Mechanism of action of SNF472, a novel calcification inhibitor to treat vascular calcification and calciphylaxis. Br J Pharmacol. 2020 https://doi.org/10.1111/bph.15163
. . .
